Fortera®
Fortifying Osteoconductive Biocement
Combines industry leading compressive strength with superior flow characteristics.

Product Overview
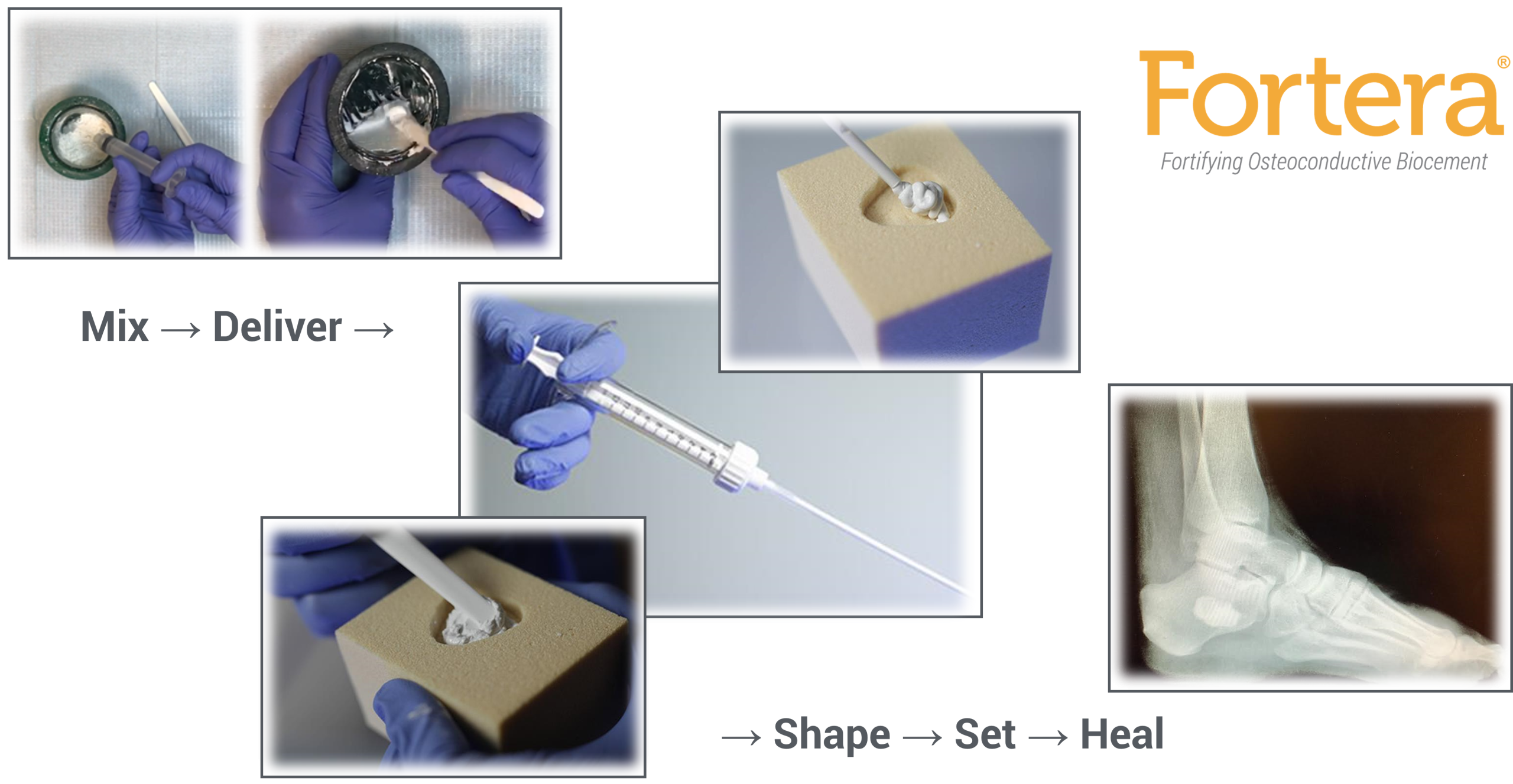
Fortera is a biocompatible, self-setting, gradually resorbable, calcium phosphate cement, supplied in a sterile, single use kit. After briefly mixing the product components at the point of use, the material transforms into a paste, capable of flowing through a 16 gauge cannula. Fortera has a working time up to 7 minutes, after which the material will begin to set. Upon setting, the material converts to hydroxyapatite, the mineral composition of native human bone.
Fortera is resorbed and remodeled by the body as new bone formation occurs during the healing process.
Fortera is intended for use to fill bony voids or defects of the skeletal system (i.e. extremities, pelvis) that may be surgically created or osseous defects created from traumatic injury to the bone.
Superior Compressive Strength†
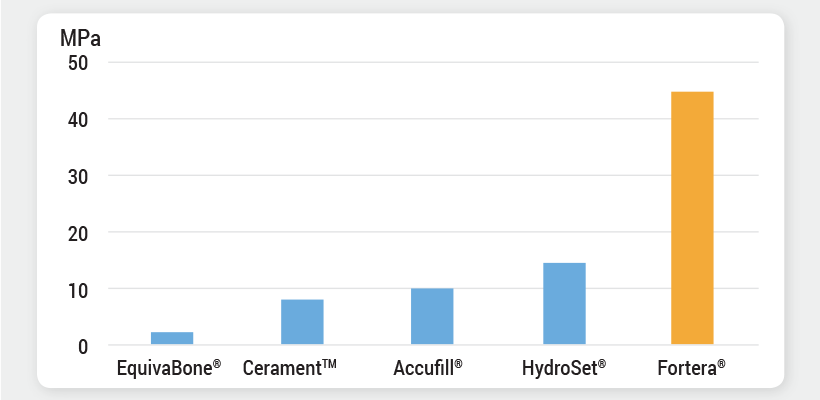
† Fortera is a trademark of Vivorté, Inc. All other trademarks are the property of their respective owners. Compressive strength reference values obtained from the following sources: Colon et al. The Knee (2015); Wang et al. Journal of Orthopaedic Translation (2016); EquivaBone Bone Graft Substitute Product Brochure, Zimmer Spine (2015), Vivorté data on file.
Performance Characteristics
| Composition | Fully synthetic |
|---|---|
| Compressive Strength | 45 MPa |
| Injectable | 16 gauge |
| Osteoconductive | Material crystallizes into hydroxyapatite upon setting, the mineral phase of native human bone |
| Thermal stability | Does not require refrigeration prior to use |
| Isothermic setting | Solidifies without generating heat, eliminating potential for thermal damage |
| Resistant to lavage | Solidifies in a wet field |
| Sizes | 3cc, 5cc, 10cc |
A 3.5mm outer diameter x 150mm length cannula is provided in each kit. Refer to package insert for additional product and safety information.
GFS-00032-05
